Producers of local intravenous products are asking the Ghana Health Service to move to stop using imported intravenous products.
They argue that such products pose serious health risks to patients.
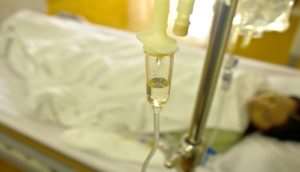
Intravenous products or IV, are health products such as fluids, drugs or blood that are administered through infusion directly into the circulatory system through a vein.
Although some of these products are manufactured in the country, the majority of products used by most facilities across the country are imported.
A Director of a local manufacturing company, Intravenous Infusions PLC, Professor Gladys Amponsah has said the quality of the imported products may not be easy to determined due to the scanty information available.
She said the safest plan is for Ghana's health agencies to source the product from local manufacturers whose productions are checked by regulators.
“These imported products, you have no idea how they are produced, what quality assurance measures are put in place or who the regulators are whereas the products Intravenous Infusions produces we know because we have a Quality Assurance manager, we have our regulators who come to the factory every year to inspect, give their recommendations and what they are not happy about and we comply.”
Professor Gladys Amponsah also argued that although the imported products may be cheaper than those manufactured locally, it can cause problems for the patients who may end up receiving ineffective products.
“The price is key for everyone. You can give a cheaper product to a patient but the consequences may be more expensive to the patient,” she warned.
Meanwhile, Intravenous Infusions PLC has delivered a strong performance in 2018 with revenue up by over 23.4 per cent to GH¢20.21 million compared to GH¢16.37 million in 2017.
Mr David Klutse, the Managing Director, Intravenous Infusion PLC, said the operating profit after tax for the year was up four per cent to GH¢2.84 million compared to GH¢2.73 million in the previous year.
Mr Klutse speaking at the Company's Annual General Meeting in Accra said the reports shared with stakeholders indicated that they were firmly on track and delivering on their Initial Public Offer (IPO) commitments.
He said the Company has also identified six strategic priorities that they believed would drive their success.
“We are taking steps to build greater effectiveness and efficiency into how we work and in manufacturing, we are adjusting production capacity to match our evolving product mix,” he added.
Mr Klutse said the progress, made in the year under review highlights the benefits of the strategic choices they had made in the Company over the last few years to build sustainable increased capacity.
“We expect the Company to make positive progress in the coming year as we are positioned to deliver strong returns to our valued shareholders,” he added.
---citinewsroom




 2024 elections: Resign if you can't be faithful to party - Sagnarigu NDC PC desc...
2024 elections: Resign if you can't be faithful to party - Sagnarigu NDC PC desc...
 Five arrested, remanded over alleged murder of two police officers at Transacco
Five arrested, remanded over alleged murder of two police officers at Transacco
 Tax exemptions better than incentives for churches – Tax Analyst tell Bawumia
Tax exemptions better than incentives for churches – Tax Analyst tell Bawumia
 Transport Minister sues Law Platform Editor for defamation
Transport Minister sues Law Platform Editor for defamation
 Voter registration: Police arrest NPP Treasurer for Mpohor for registering minor
Voter registration: Police arrest NPP Treasurer for Mpohor for registering minor
 "This nonsense must stop" — Lawrence Tetteh vows to march to Jubilee House over ...
"This nonsense must stop" — Lawrence Tetteh vows to march to Jubilee House over ...
 2024 elections: “If indeed you stand for peaceful elections the time is now for ...
2024 elections: “If indeed you stand for peaceful elections the time is now for ...
 I have the attributes to be president of this country — Bernard Monarh
I have the attributes to be president of this country — Bernard Monarh
 Cecilia Dapaah saga: ‘Turf war’ between AG, EOCO, OSP indicates they’re not ‘cor...
Cecilia Dapaah saga: ‘Turf war’ between AG, EOCO, OSP indicates they’re not ‘cor...
 Ghana will become the first African country to embrace blockchain-powered gover...
Ghana will become the first African country to embrace blockchain-powered gover...
