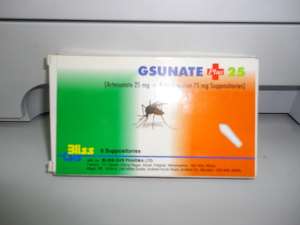
The anti-malarial, a suppository made up of combination of Artesunate 25mg and Amodiaquine 75mg, which was manufactured in India by a company called BLISS GVS PHARMA Limited, has not undergone any Clinical Trial study on its combination.
A release issued and signed by the Chief Executive of FDA, Dr Stephen Opuni, copied to the Ghana News Agency, said Gsunate Plus suppository was not registered by the FDA and the drug could not be guaranteed for the treatment of malaria in children.
'The efficacy of the combination of artesunate and amodiaquine through the rectal route has not been established and therefore treatment of malaria in children with this drug could lead to therapeutic failures with complications', it said.
It instructed hospitals, clinics, pharmacies, licensed chemical sellers and other health facilities having stocks of GSUNATE Plus Suppository to immediately stop dispensing them and handover stocks to the nearest FDA office countrywide for safe disposal.
It said the importer; Tobinco Pharmaceutical Limited was assisting the Food and Drugs Authority (FDA) to recall the drug from the market.
The release however called for any information from the general public on persons in any practice possible of endangering public health and safety with respect to FDA's mandate, adding that, 'The general public should call the numbers; 0244337235, 0544863418, 0244571563 or 0244337250 to release any information'.
GNA/Ghana




 NaIMOS arrests 49 suspected illegal miners after armed clash at Adiewoso
NaIMOS arrests 49 suspected illegal miners after armed clash at Adiewoso
 Upper East Regional Minister leads technical assessment for proposed airport pro...
Upper East Regional Minister leads technical assessment for proposed airport pro...
 Missing car dealer found dead as police arrest friend over alleged murder
Missing car dealer found dead as police arrest friend over alleged murder
 Teacher Sir Ola shares inspiring story of sacrifice and compassion
Teacher Sir Ola shares inspiring story of sacrifice and compassion
 Extreme heat is a growing threat to health, jobs and food security in southern A...
Extreme heat is a growing threat to health, jobs and food security in southern A...
 Mali lawyer critical of junta abducted overnight
Mali lawyer critical of junta abducted overnight
 Two women die on migrant boat near France seeking to reach UK
Two women die on migrant boat near France seeking to reach UK
 Iran offers strait deal, Trump says he's not satisfied
Iran offers strait deal, Trump says he's not satisfied
 How faulty fan regulator sparked fire, destroyed two rooms at Suhum Konfine
How faulty fan regulator sparked fire, destroyed two rooms at Suhum Konfine
 Don’t store bread beyond four days – Baker advises
Don’t store bread beyond four days – Baker advises

