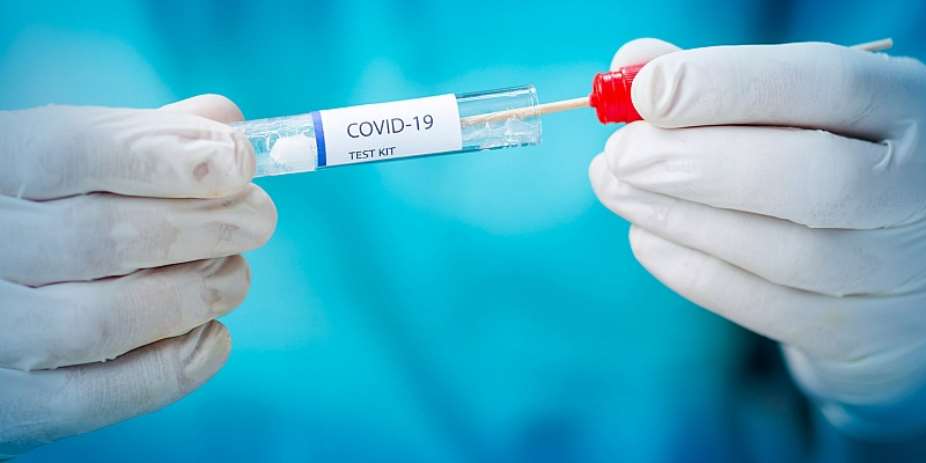
Should not a medicine (Remdesivir) that has shown lifesaving effect in certain cases of Covid-19 be made available to all those who need it without delay? Even if Big Pharma has a patent, there are provisions in the global trade treaties that allow governments to issue compulsory licenses to such a lifesaving drug, and keep people over profit. This is why medical experts are demanding governments to use compulsory licensing for generic production of such a drug to help save lives.
Compulsory licensing is one of the key public health and social justice safeguards that allows governments to use, produce, and import or export patented technologies in the public interest.
When confronted with intellectual property barriers that block access to affordable lifesaving treatments, governments of countries like Thailand, Brazil, India, South Africa, Malaysia among others, have used this mechanism of compulsory licensing in the past, to ensure lifesaving medicines reach the people who need it most (for HIV, hepatitis C, cancer, etc). Compulsory licensing enables local production or importation of generic medicines, multiple supply options, reduces price and increases access to lifesaving treatments for those most in need.
"Though Indian state of Maharashtra is registering high numbers of Covid-19 cases as of now, situation is equally bad in several states. Among the medicines that have shown to work in Covid-19 are steroids and Remdesivir . The patent owner of Remdesivir, Gilead, had given voluntary license to six Indian pharmaceuticals. Even then, there is an acute shortage of Remdesivir injections, possibly due to hoarding and racketeering, especially in Maharashtra. This will have cascading effects in rest of India. Its maximum retail price ranges between INR 2800-5400 (as per the company that produces it) whereas procurement rates at hospitals range between INR 600-1000. After we pointed out this discrepancy, the Maharashtra government has once again capped the price from INR 1000 to INR 1400 per injection. Each eligible patient needs six doses and required only for treating moderate or severe Covid cases" said Dr Ishwar Gilada, Secretary General of Organized Medicine Academic Guild (OMAG), an umbrella network of several professional medical experts' associations in India.
OMAG has appealed to the Indian Prime Minister to put Remdesivir in Drug Price Control Order (DPCO), as is done for other life-saving medicines. This will help reduce the maximum retail price to affordable level and remove buffer margins and scope for black-marketing.
Another ask of OMAG is to grant a compulsory license under Section 84 of Indian Patents Act, 1970, to ramp-up Remdesivir production. It can bring-down procurement cost to below INR 500 per vial, to save millions of Indian Covid patients as also several more globally, who will benefit from made-in-India generic Remdesivir.
OMAG has also insisted on rationalizing use of Remedesivir with strict adherence to the guidelines, making multiple Remdesivir stores or Remdesivir Banks with strict sales control, and making racketeering of Remdesivir punishable under the Epidemic Act of India.
Dr Ishwar Gilada who is also the elected President of AIDS Society of India and Governing Council member of International AIDS Society (IAS), added that Remdesivir (GS-5734) was originally developed by Gilead Sciences in 2009, to treat Hepatitis C and respiratory syncytial virus (RSV), but failed. It was then repurposed and studied as a potential treatment for Ebola and Marburg virus infections. In collaborative study it was subsequently discovered that it had antiviral activity in vitro against multiple filoviruses , pneumoviruses , paramyxoviruses, and coronaviruses . It is now repurposed for use in Covid-19. Remdesivir was granted patent in India vide No. 7068/DELNP/2010 (IN275967) as Markush type claims that allow important innovations to be patented and No. 7404/DELNP/2010 (IN289041) as Molecule in 2010. A compulsory license can be issued against any drug with three year old patent and hence this demand from OMAG.
One of the lessons from the global public health emergency, is that profiteering from illness has to end. It is an essential cog-in-the-wheel of sustainable development where no-one-is-left-behind that all healthcare services reach every human being in a rights-based manner - and - not dependent on her or his capacity to pay.
Médecins Sans Frontières (MSF or Doctors Without Borders) had earlier stated that a compulsory license is a license for alternative production or importation of a generic version of a patented medicine which is granted by the government and does not require the consent of the patent-holder. The Doha Declaration on TRIPS Agreement and Public Health confirms that countries are free to determine the grounds of compulsory licenses. As we know, TRIPS is Trade-Related Aspects of Intellectual Property Rights, which is an international legal agreement between all the member nations of the World Trade Organization (WTO). Examples of different grounds for compulsory license include, for instance to remedy anti-competitive practices, failure to work or insufficient working of the patent, when the patented medicine is unaffordable or unavailable making it inaccessible to patients and when public health is at stake including but not limited to emergency/extreme urgency, epidemics and public non-commercial use.
Bobby Ramakant – CNS
(Bobby Ramakant is a 2008 World Health Organization (WHO) Director General’s WNTD Awardee and part of CNS (Citizen News Service), Asha Parivar, and Socialist Party (India). Follow him on Twitter @bobbyramakant or read www.bit.ly/BobbyRamakant)
- Shared under Creative Commons (CC)




 Assemblyman raises alarm over alleged illegal ECG charges in Ho
Assemblyman raises alarm over alleged illegal ECG charges in Ho
 GIS defends Asikuma–Sogakope checkpoints, cites security and border management
GIS defends Asikuma–Sogakope checkpoints, cites security and border management
 Dr. Zenator Rawlings elected 2nd Vice President of Pan African Parliament
Dr. Zenator Rawlings elected 2nd Vice President of Pan African Parliament
 Fake online businesses dupe victims of GHS266k between January and April — CSA
Fake online businesses dupe victims of GHS266k between January and April — CSA
 BoG's 2025 spending was necessary in saving a near collapse economy — Majority
BoG's 2025 spending was necessary in saving a near collapse economy — Majority
 Ghanaians need solution to the current dumsor, not your excuses — Oforikrom MP t...
Ghanaians need solution to the current dumsor, not your excuses — Oforikrom MP t...
 NACOC intercepts GHS5million tablets of Tapentadol in major drug bust
NACOC intercepts GHS5million tablets of Tapentadol in major drug bust
 Oti Region: Police arrest two over alleged illegal possession of firearm
Oti Region: Police arrest two over alleged illegal possession of firearm
 Victor Smith engages Boeing on rebirth of Ghana Airways
Victor Smith engages Boeing on rebirth of Ghana Airways
 We were told dumsor would end with the levy – Dr Zaato fumes
We were told dumsor would end with the levy – Dr Zaato fumes

