
Practitioners of Homeopathy & alternative medicine who do not abide by the rules of evidence-based practice may fall foul of the law ( Edzard Ernst , 2012).
Being LLB Law student and a practitioner, researcher and promoter of evidence-based alternative and holistic medicine, MY FAVOURITE law CASE in contract was the Michael V O’MahonyAn 1890s ad that claimed a product would prevent a person from contracting flu. The case is still a headline one for showing advertisers the kind of claims they should never make. Interesting, the carbolic smoke ball was a homoeopathic product produced by a homoeopathic company which was marketed aggressively to fight the flu pandemic then. This case is still relevant today in the recent coronavirus pandemic declared by the World Health Organization(WHO).

The fact of the case:
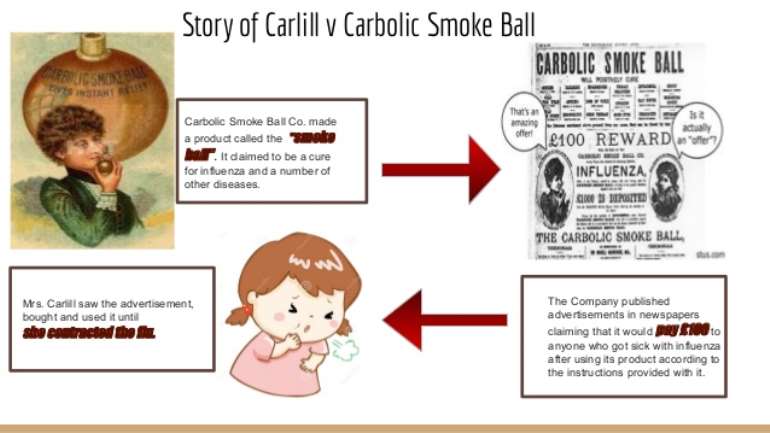
The 1892 case of Carlill v Carbolic Smoke Ball Company, which related to an advertisement placed in the Pall Mall Gazette promoting the use of the defendant’s carbolic smoke ball to prevent a person from contracting influenza, and promising £100 to any user who did get the flu is a locus classicus in law of contract.
The facts of the case conjure up a Punch-like cartoon image of a Victorian lady, head covered with a towel, leaning over and breathing in waves of carbolic smoke fumes. The actual text of the advert is part of what makes this case amusing. Every element of it was analyzed during the case. As well as promising a £100 reward, the company also said that £1,000 had been deposited with Alliance Bank of Regent Street “showing our sincerity in the matter”. It also said: “During the last epidemic of influenza many thousands of carbolic smoke balls were sold as preventatives of this disease, and in no ascertained case was the disease contracted by those using the carbolic smoke ball.
“One carbolic smoke ball will last a family several months, making it the cheapest remedy in the world at the price, 10s. post free.” On the faith of this advert, Mrs Carlill bought and used the smoke ball as directed for two months, but in January 1892 nevertheless did contract the flu. She then bravely embarked on a claim for the £100 promised in the advert – £100 in those days being a very tidy sum indeed.
The defendants unsuccessfully argued that the advert was a mere “advertising puff”, which was never intended to create a legal relationship with any individual purchaser of a smoke ball in reliance on it.
Secondly, they argued that if a contract was thereby created, it was void as a wagering contract. They also put forward the argument that it was an illegal form of contract insurance.
However, Mrs Carlill’s fortitude paid off – she succeeded in both the English high court and court of appeal, and recovered the £100 plus her legal costs. Both courts held the reliance by Mrs Carlill on the advert established a contract by the defendants to pay her £100 in the event that happened.
Why is this my favorite case?

The Carlill case became my favorite partly because it falls under the jurisdiction of false claims and the company in question was a homeopathic company. It signifies the many advertisements and claims made by practitioners of herbal, homeopathy and alternative medicine. The sui generis is that it exemplified a number of legal principles relating to the formation of a contract, which can be expressed in simple terms as an offer by one party that is accepted by another where something of value (ie consideration) passes between the two parties.
Not too many cases exemplifying contract principles have a human face on them, but the Carbolic Smoke Ball Company case did. I found that, the facts and imagery of this case enlivened proceedings and changed my position on the kind of claims to put out there to the public. In fact, studying law change my posture in the practice of holistic medicine and I look at my practice and the law from a different perspective. In fact, I now issue disclaimers to all my articles in my column and my natural products formulated.
I believe practitioners of homeopathy and alternative medicine would want this case to redraft the advert in a way that would extol in a most persuasive way the alleged efficacy of the smoke ball but without the risk of it constituting an offer to the whole world capable of creating a contract with someone using it.
Is this case still relevant?
Yes! This is similar to the carbolic smoke ball case which killed millions. The Carlill case, more than any other “promise to the world”-type case, is the headline case in terms of showing advertisers where not to go and if practitioners of homeopathy and alternative medicine do not take care, we could have a new modern case in this times.
One of the appeal court judges, Lord Justice Lindley, put it succinctly: “It appears to me, therefore, that the defendants must perform their promise, and, if they have been so unwary as to expose themselves to a great many actions, so much the worse for them.”
To avoid that risk, advertisers, while still engaging in what has been described as “the eulogistic commendation of the res vendita” (ie using merely laudatory commendation of what is being sold), do not actually make binding promises about the product being promoted. This is very important to the alternative medicine industry.
How often do we hear spot adverts on the radio expounding the virtues of a particular product but then concluding in a quieter tone that “terms and conditions apply”? This is to ensure that an unconditional and open commitment is not made to a listener who purchases the product on the strength of what is said.
Remember, in most instances nowadays it is still a case of caveat emptor (let the buyer beware). The formidable Mrs Carlill was a notable exception.
Consumer case law and complementary medicine

A locus classicus dealing with misleading or deceptive behaviour relevant to complementary medicine is ACCC v Purple Harmony Plates Pty Ltd. It had to do with representations made in relation to a product called ‘Purple Harmony plates’, which came in different forms including disks, angels, phone disks and fridge fresheners. These products were said to have many and diverse therapeutic benefits, including: negating the effects of electromagnetic radiation; accelerating healing; calming people; increasing health; decreasing stress levels; strengthening the immune system; treating cuts, burns, aches and pains; green thumbs to treat water and a fuel ionizer system to treat fuel and improve its efficiency.
The court held that the defendant had represented the products possessed the performance characteristics claimed and that these representations made claims as to future matters and suggested that a person who purchased the product would derive the stated benefits from the product. The defendant did not provide any substantial evidence to support the assertions made that would address the question of whether the company had any reasonable grounds for making the representations. Accordingly, the representations were deemed to be misleading. The court ordered injunctive relief against the respondents making these representations and required refunds to customers and corrective advertisement. Similar issues arose in Commissioner of Fair Trading, Department of Complementary and alternative medicine and consumer law.
Also in Commerce v Perrett, the Commissioner pursued a declaration that the respondent Perrett had betrothed in misleading or deceptive conduct and conduct likely to mislead or deceive in contravention of the Fair Trading Act 1987 (NSW). It was also suggested he had engaged in conduct that was liable to mislead the public as to the nature, characteristics and suitability for their purpose of certain goods in contravention of s 49 of the same legislation. The Commissioner sought a restraining order in relation to these types of activities. The court dealt with the defendant’s actions in relation to a number of clients, involving the use of unorthodox substances of uncertain or unknown composition, the diagnosis and treatment of acute and chronic medical conditions and the application of ointments and intravenous injections. The Commissioner suggested that the defendant had represented he had an ability to treat multiple sclerosis, breast cancer, a thyroid condition, terminal cancer, Huntington’s Disease and sympathetic nerve dystrophy.
The Commissioner also argued that the defendant made statements suggesting clients should not rely upon medical treatment and indicating he had access to knowledge and substances not normally available. The defendant was not able to present evidence to support the representations he made about his ability to treat those ailments. The plaintiff relied upon s 41(2) that placed the burden of proof upon a person who has made a representation about a future matter to demonstrate that he or she had reasonable grounds for making the representation, otherwise the representations would be deemed to be misleading. The court relied upon statements of clients, and on that basis Perrett was held to have made representations that were misleading or deceptive or likely to mislead or deceive; restraining orders were granted to stop that behaviour. In the same year as the Perrett decision, misleading representations in breach of the Trade Practices Act were held to have been made by the respondent company in ACCC v NuEra Health Pty Ltd (in liq).

The statements in question, which promoted NuEra’s products and ‘treatments’ to cancer victims via the company’s website, were categorised by the ACCC as the ‘cure cancer representations’, the ‘prolong life representations’ and the ‘scientific support representations’, and condemned by Ryan J as exemplifying ‘conduct of the most reprehensible kind’. In coming to this decision, his Honour was scathing of the fact that the respondent had not adduced any evidence to contradict or ‘palliate’ the case presented by the ACCC.
In ACCC v Jones, the ACCC instituted proceedings against Darryl Peter Jones in respect of alleged contraventions of the Trade Practices Act. The ACCC claimed that a website Mr Jones maintained and a publication authored by him, entitled ‘The Truth about Overcoming Cancer’, contained representations that were misleading or deceptive:
The Darryl Jones Health Resolution Centre methodology is based on the resolution of life-threatening diseases without dispensing pharmaceutical drugs, advocating radium therapy, surgery, or harmful chemotherapy — focusing instead on a three-step Triune Wellness Offensive — utilizing nutrition, exercise, and vitamins, along with close, professional, personal accountability.
On behalf of the ACCC, physician Dr Snyder FRACP indicated that there was either limited or no scientific evidence for these types of representations. On the balance of this evidence, there was a finding by the court that the statements were misleading or deceptive.
In ACCC v Advanced Allergy Elimination Pty Ltd, an action was commenced by the ACCC against the respondent Advanced Allergy Elimination (AAE), which operated clinics for the diagnosis and treatment of allergies. The methods by which AAE diagnosed and treated allergies included testing for and identifying a person’s specific allergies using a muscle strength indicator technique and treating allergies by using a technique based on positive and negative conditioning.
The assumption appeared to be that, as there was limited scientific evidence for the statements, accordingly they were misleading or deceptive.
In a analogous factual circumstance in ACCC v Willesee Healthcare Pty Ltd, the court determined that representations by the respondent about treating allergies through kinesiology and acupressure and thereby being able to cure or eliminate all, or virtually all, allergies or allergic reactions were misleading and deceptive representations, also based upon the expert evidence of Professor Douglass. This conclusion was reached in the absence of other evidence to the contrary.
In Commissioner of Fair Trading, Department of Commerce v Hunter, an injunction was sought against Hunter, who practiced as a naturopath and medical herbalist. The primary focus of the case was in relation to his advertising of ‘live blood analysis’, which was said to allow diagnosis of ailments instantly and to assist in the treatment of such illnesses that the Commissioner suggested was a misleading representation under the terms of the Fair Trading Act. Also of concern in this matter were the representations made about the qualifications of Mr Hunter and his ability to diagnose and treat serious health conditions. Mr Hunter used the titles ‘Dr’, ‘Doctor of Natural Medicine’ and PhD in advertisements as well as words that could suggest he was a medical doctor. As Mr Hunter was not a medical doctor, these representations were considered to be misleading or deceptive representations. Also deemed to be misleading or deceptive was the representation that he was competent to treat serious illnesses such as high blood pressure and a list of other conditions. Evidence was presented from Professor Eva Raik, a haematologist with extensive qualifications and experience about the efficacy of live blood analysis and the difficulty in ascertaining medical conditions or making therapeutic decisions or diagnosis from that type of study. There was limited evidence produced by Hunter in relation to the evidence basis for this therapy. The court found, on the basis of the expert evidence, that the representations in regard to the value of live blood analysis were misleading or deceptive or likely to mislead or deceive.
The court ordered that Mr Hunter be permanently restrained from carrying on a business or in any way providing in trade and commerce naturopathy, medical herbalism, herbalism, iridology, hydrotherapy, sports medicine, osteopathy and blood analysis.
In another case involving, ACCC v Giraffe World involved the marketing of an ‘ion mat’ (the mat) by the first respondent Giraffe World. The mat or mattress was connected to a source of electricity. There is nothing in the judgment to suggest this item had been listed as a therapeutic device under the Therapeutic Goods Act. The ACCC alleged that Giraffe World made misrepresentations in contravention of ss 52 and 53(c) of the Trade Practices Act, in suggesting that as a result of its emission of negative ions the mat benefited the health of persons who slept on it. Giraffe World provided some expert testimony to support these assertions, including evidence from an experienced naturopath, a chiropractor and an academic engineer.
There was also reference to a published article by a Japanese professor about the impact of these types of equipment on the human body, though without suggesting any particular health benefits. The ACCC presented evidence from a group of medical doctors and university experts in physics about the performance and impact of the mat. The judge concluded that the burden of the proof provided by the ACCC suggested he should prefer the view of the ACCC, and this resulted in the court determining the representations were misleading or deceptive. This case process further entrenches the view that statements made about a piece of equipment should be supported by scientific evidence, otherwise there is a possibility of a finding of a breach of consumer legislation. A significant authority that deals directly with the issue of what type of evidence is required to avoid a finding that an activity is deemed misleading or deceptive is Noone, Director of Consumer Affairs Victoria v Operation Smile. In this case, Four respondents (collectively, Operation Smile) operated the ‘Hope Clinic’, described as a complementary medicine centre specializing in the treatment of cancer. Operation Smile’s website described the treatments offered at the Hope Clinic for a long list of serious medical conditions such as cancer and HIV and contained statements relating to the efficacy of these treatments based upon peer reviewed and published methods of cancer treatment; it claimed that the techniques used at the clinic included state-of-the-art medical technology and subtle energy therapies such as homoeopathy and acupuncture.
The Director of Consumer Affairs alleged that Operation Smile engaged in misleading or deceptive conduct in trade or commerce contrary to s 9(1) of the Fair Trading Act 1999 (Vic)on the basis that the statements falsely represented that the treatments offered by Operation Smile treated cancer effectively based upon scientific evidence.
The Court of Appeal held the net effect of the statements was that the treatments offered were as scientifically based and rigorously tested as those of conventional medicine based upon an assessment of the whole of the context within which the statements were made.
As appears from what is set out above, the context included the claim that the Hope Clinic adopted an integrated approach to the treatment of chronic illness combining state-of-the-art medical technology with alternative therapies; the explicit description of Hope Clinic therapies as the best scientific complementary medicine; the ostensibly scientific names ascribed to the therapies offered by the Hope Clinic, such as ‘photo dynamic therapy’, ‘Holt Microwave therapy’ ‘Oxygen therapy’ and ‘Biolife electrotherapy’; the assertion as to practitioners of complementary medicine not hesitating ‘to employ conventional medical practices’; the explicit assertion that the Hope Clinic’s combination of alternative treatments with conventional medicine would optimize a patient’s treatment plan; and the surely very remarkable claim that: The network of colleagues who make up this institute are committed to reading and researching all published information. We contact scientists and physicians all over the world to learn at first hand from these eminent colleagues in hospitals, universities and laboratories.
Although the Court of Appeal allowed the appeal and held that the statements discussed in the case were misleading or deceptive, this decision is significant in that it indicated that, if precision is applied to statements made which provide clarity for the reader about the basis of the evidence to support the statements made that a breach of the misleading or deceptive conduct prohibition might be avoided.

Homeopathy & Alternative Medicine and the Law.
So in the course of studying law, I realized that, Homeopathy and Alternative medicine practitioners can't escape the long arm of the law. Practitioners who offer unscientific treatments are acting unprofessionally, regardless of whether they get patients' informed consent. Alternative medicine has been in the firing line for a very long time. By and large, the critics are healthcare professionals who argue that the therapeutic claims made for alternative treatments are neither evidence-based nor plausible.
In recent years, their arguments have been increasingly adopted by the legal profession and the battles over alternative medicine are increasingly fought in the courts. In the UK, Simon Singh famously won the libel case brought against him by the British Chiropractic Association. In the US, a patient has accused Stanislaw R Burzynski, a proponent of alternative cancer cures, of swindling her out of nearly $100,000 (£63,000) by using "false and misleading tactics". The case is ongoing. Also in the US, a woman was awarded $7.4m (£4.7m) after suffering a stroke following the intake of an herbal supplement.
And this is the big deal, the world's largest homeopathic manufacturer Boiron recently settled for CAD$12m (£7.4m) after a class action that was brought against it in Canada for selling homeopathic remedies under false pretenses)
Dealing with CAM in Consumer law.
With regards to claims of misleading representations in advertising or in the course of practice by health practitioners, the case law suggests that these claims are evaluated based upon the need for high quality scientific evidence. According to Weir et al 2013, “This is no doubt reflective of a view that when dealing with health matters there is a need for good evidence to avoid personal injury.
Recommendation
It is not intended that this article should provide a counsel to not require an adequate evidence base for representations made about CAM. There are justified concerns expressed by many about the outrageous representations made by some unregistered health practitioners, and it is appropriate that these practices are dealt with.
In negligence cases against CAM practitioners, the evidence of expert CAM practitioners is accepted as influential evidence even in the face of other contrary Orthodox Medicine expert evidence-Shakoor v Situ.
This issue arises under the claims guidelines, which allow the use of traditional evidence to justify certain types of representations. A similar level of evidence should be accepted in the case of CAM advertising and representations generally and in effect apply some of the processes applied by the Australian Register of Therapeutic Goods process more widely. In the case of advertising or representations in regard to curing or treating serious illnesses, there is a basis to argue for some limitations on representations in that circumstance requiring high quality scientific evidence.
Conclusion
Komesaroff et al 2012 have suggested that, rather than suppressing all approaches to health care that we cannot understand or condone:
[A] system of safeguards should be established to minimize risk, while continuing to protect the rights of consumers to choose their own health-care practices. Such safeguards should include legal, professional and conceptual criteria and target specific rogue practices while protecting and regulating others. Medicine is a complex craft and much of its success depends on its ability to draw on a wide array of practices . . . We cannot afford to be overconfident about our own approaches or dismissive of those of others.
So in essence, this tells us that the practitioners, promoters or manufacturers of alternative medicines who do not abide by the rules of evidence-based practice are likely to be operating outside the law. This makes a lot of sense. How much of alternative medicine would survive if the legal principles were rigorously applied in Ghana?
Well, we can let the discussion begin on homeopathy, alternative and herbal medicine and the law in this coronavirus pandemic and aggressive marketing from the practitioners. It is time for the government to bring sanity in the practice of homeopathy, alternative and herbal medicines in this hard times. Those who claim to have any product should be able to present their products to scientific analysis instead of using the media to propagate their false claims.
DISCLAIMER : This post is for enlightenment purposes only and should not be used as a replacement for professional advise.
Dr. Raphael Nyarkotey Obu, PhD, Is an honorary Professor of Naturopathic Medicine with research interest in Naturopathic & Holistic Urology, Vinnytsia State Pedagogical University, Ukraine. President of Nyarkotey College of Holistic Medicine- Formulator of FDA approved Men's Formula for Prostate Health, Women's Formula for wellness & Nyarkotey Tea for cardiovascular Health. 0241083423 or 0541234556. The author also is currently pursing LLB Law/MBA concurrently





 GJA Applauds Ghana’s Sharp Rise in Global Press Freedom Rankings
GJA Applauds Ghana’s Sharp Rise in Global Press Freedom Rankings
 US Embassy Cautions Against Censorship in Fight Against Misinformation
US Embassy Cautions Against Censorship in Fight Against Misinformation
 Interior Minister Blames Weak Enforcement by Assemblies After Avenor Building Co...
Interior Minister Blames Weak Enforcement by Assemblies After Avenor Building Co...
 Gov’t Warns Against Rising Misinformation, Calls for Stronger Journalistic Stand...
Gov’t Warns Against Rising Misinformation, Calls for Stronger Journalistic Stand...
 Ramaphosa Warns Against Vigilante Crackdowns on Foreign Nationals
Ramaphosa Warns Against Vigilante Crackdowns on Foreign Nationals
 Global InfoAnalytics Boss Rejects Claims Polls Are Destabilising NDC
Global InfoAnalytics Boss Rejects Claims Polls Are Destabilising NDC
 Bawumia to Propose Policy Alternative as Cocoa Sector Tensions Deepen
Bawumia to Propose Policy Alternative as Cocoa Sector Tensions Deepen
 ECG Announces Scheduled Outages and Technical Fault Affecting Multiple Regions o...
ECG Announces Scheduled Outages and Technical Fault Affecting Multiple Regions o...
 Investigation committee uncover GH¢19.5m loss at Bolgatanga Technical University...
Investigation committee uncover GH¢19.5m loss at Bolgatanga Technical University...
 Afenyo-Markin calls for protection of journalists, warns against suppression of ...
Afenyo-Markin calls for protection of journalists, warns against suppression of ...

